|
It is also called wood alcohol because it can Ethanol is the alcohol found in alcoholic beverages. Ethanol isĪlso known as grain alcohol because it can be obtained from the fermentation of grains (rye,Ĭorn etc.) which contain starch. The functional group for alcohols is the -O-H group. The single bonds account for 16 of them the remaining two pair are accommĮthanol, CH 3CH 2OH, is also called ethyl alcohol and it is the second member of the class ofĬompounds known as alcohols. In this case there are 20 valence electrons: 6 from 6 hydrogens, 8 from 2 carbons, and 6 from Purpose because it has been found to be very toxic. Molecule looks more like the first than the second.Ĭarbon tetrachloride used to be used as a "dry cleaning" spot remover. Involve charge separation and the second does, the first is the greater contributor, i.e. The two alternatives are the resonance structures for this compound. The calculations of formal charge for the oxygen and chlorin Reside, unshared, on the chlorine, and six, unshared, on the oxygen. This takes care of all 12.Īlternatively, if we accommodate two of the remaining 12 valence electrons between theĬhlorine and carbon to which it is attached, forming a second bond there, then 4 electrons can Oxygen, and six, unshared, on the chlorine. Nd there, then 4 electrons can reside, unshared, on the Which it is attached, forming a second bo If we accommodate two of the remaining 12 valence electrons between the oxygen and carbon to (Two resonance structures the one which involves charge separation does not make a large 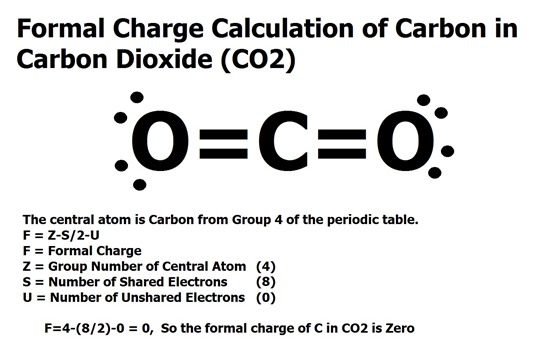 
Alkynes are hydrocarbons that have a triple bond. Alkenes are hydrocarbons that have a double bond. 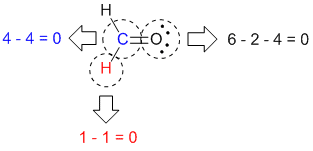
Ompound which consists of only hydrogen and carbon atoms. An alkane is a hydrocarbon in which there are no multiple bonds. Lewis structure in these cases the existence of resonance will be indicated.įrom this point on the calculation of formal charge will be shown only if the result is not In some cases there will be more than one reasonable > distributing those electrons on the structure in an appropriate way, andīe sure to show all unshared electrons. > adding up the number of valence electrons, Draw Lewis structures of the molecules below.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
